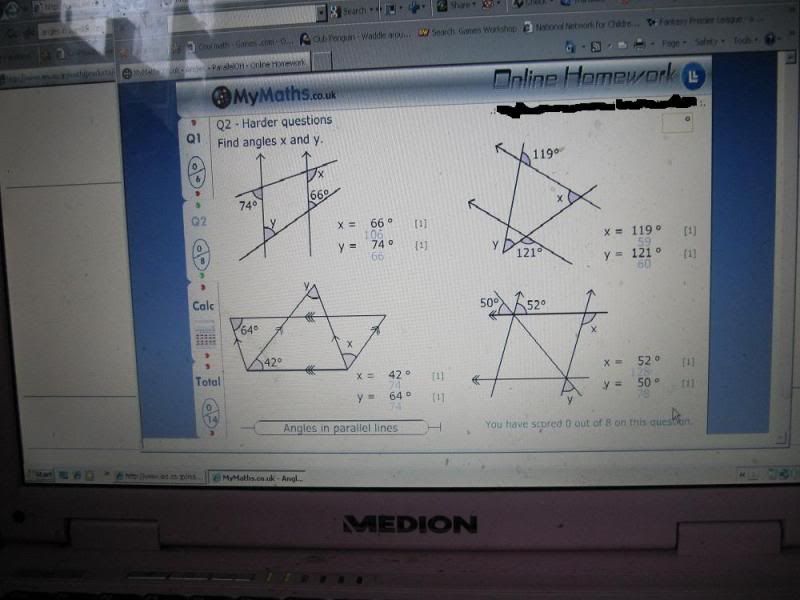
Essay on Water for Children and Students.
Hydrogen bonds hold neighboring water molecules together. This cohesion helps the chain of water molecules move upward against gravity in water conducting cells as water evaporates from the leaves Adhesion between water molecules and the walls of the water conducting cells also helps counter gravity.
The Biological Significance Of The Properties Of Water The human body 15% lipids, 12% proteins but by far the biggest % is water which makes up a massive 70% of the human body. This is the equivalent of 49kg in an average male adult human of mass 70kg. This is a good indication of just how important.
Previous IB Exam Essay Questions: Unit 3 Use these model essay question responses to prepare for essay questions on your in class tests, as well as the IB Examination, Paper 2. These questions have appeared on recent IB examinations, exactly as shown below. Following each question is the markscheme answer which was used to evaluate student.
Importance of water in our life Water is the most important substance in the world, a necessity utilized by living things for survival but also necessary for several processes. Water is vital in our life due to the following reasons. It is essential for our survival. Experts estimate that on average a human body requires about 48 ounces of water to.
Properties of Water Water is essential for life as we know it on earth. It is used by plants and animals for basic biological processes which would be impossible without the use of water.The origin of all life can be traced back to the water in the Earth's precambrien seas.Water is also the universal solvent. It reacts with more elements and compounds than any other substance known to man.
A basic guide on how to write an essay. Includes tips on how to write different essay types. Essay Writing Help Properties Of Water Essay. Every paper produced by our writers is unique, plagiarism free and absolutely authentic. Thank you once again for helping me with my engineering assignment. Even though it is often true, but when you come to us it will not be true at all, because we let our.
Biology essay titles. This document contains the essay titles and mark schemes used in AQA A-level Biology examinations since 2007. The specifications these exam questions came from are no longer in use, but the marking method has largely remained unchanged. Further guidance on the marking method used with the essay can be found in.
Other posts on the site.
Properties and uses of water. Water is essential to living organisms. The list below shows some of its properties and uses. Hydrogen bonds are formed between the oxygen of one water molecule and the hydrogen of another. As a result of this water molecules have an attraction for each other known as cohesion. Cohesion is responsible for surface tension which enables aquatic insects like pond.
Water's hydrogen bonds are also why its solid form, ice, can float on its liquid form. Ice is less dense than water because water molecules form crystalline structures at freezing (32 degrees Fahrenheit or 0 degrees Celsius) temperatures. The thermal properties of water are also linked to its hydrogen bonds.
Essay text: Not only does the ice absorb heat making waters cooler, but it also insulates this heat creating a livable habitat for the animals below. Evaporative cooling and high heat of vaporization are more important properties of water. Evaporation is the conversion of a liquid to a gas, while high heat of vaporization is the heat needed to.
Students will complete this Properties of Water Review to reinforce today's lesson. Sample of Student Work: Water Properties Practice Questions - Student understanding seemed to improve when students were able to demonstrate their comprehension through basic review questions. It is imperative with the NGSS to encourage students to move beyond.
This question would form part of an essay question in an examination. Exemplar question. Outline the thermal, cohesive and solvent properties of water and describe the significance of each to living organisms. (6) Student response. Water has high specific heat capacity and it can absorb or give off a lot of heat energy without changing.